This shows two layers with slightly different colours demonstrating that bromine is more soluble in non-polar solvents. Denser than water and soluble in water.
Properties Of Hydrocarbons Mendelset
Intramolecular carbonyl ene reaction.

. Cyclohexane and benzene both are the exceptions to the. Cyclohexane C6H12 Bromine Br2 -. It does not react with bromine unless energy in.
It starts with the formation of brominium ion as in direct bromination of alkenes. Ad More Available at Sigma-Aldrich. What is the type of reaction between cyclohexane and bromine in.
Explain why the addition of. Why does cyclohexane not react with. In the case of the.
Why does cyclohexene not react with bromine. Investigating the Reaction Between Bromine and Cyclohexane. In such a case a free-radical.
Bromine does not react with cyclohexane without UV light with or without water. When bromine reacts with a carbon -carbon double or triple bond or with anything else for that matter the bromine bond breaks and the bromine molecule is destroyed. The balanced equation of ethane and bromine is by double displacement.
What is product of cyclohexane react with bromine. Cyclohexene reacts with bromine in the same way and under the same conditions as any. It does not react with bromine unless energy in the form of light or heat is applied.
Science Chemistry QA Library Consider the reaction of. What is the Equation of cyclohexane and bromine. Organic yields of bromine and iodine induced by radiative neutron capture and isomeric transition in polycrystalline n-hexane and cyclohexane.
When an EAS reaction eg. Therefore Bromine test differentiates Benzene and Cyclohexane by colour change. Bromine is a dark reddish-brown fuming liquid with a pungent odor.
It does not react with bromine unless energy in the form of light or heat is applied. Cyclohexane has no pi-unsaturation and is therefore not nucleophilic. What is the type of reaction between cyclohexane and bromine in dichloromethane.
In such a case a free-radical substitution reaction occurs. Bromination is carried out on a monosubstituted benzene a mixture of ortho- meta- and para-substituted isomers forms. Cyclohexene is a typical alkene and.
By adding bromine to a mixture of Cyclohexane and water and placing. The mixture under a bright light and. If you do not know the mechanism you can refer to it here at MasterOrganicChemistry.
Expertise on every level to craft science technology solutions in life science. The mixture under a bright light and shaking from time to time Hydrogen Bromide is formed. So the correct answer is Option C.
C2H6 Br2 C2H5Br HBr Reaction equation of lithium and bromine. This reaction is shown. By adding bromine to a mixture of Cyclohexane and water and placing.
Youre welcome although you dont get bromocyclohexane because it. Cyclohexane has no pi-unsaturation and is therefore not nucleophilic. A demonstration of the reaction of cyclohexane cyclohexene and toluene with Bromine water.
No reaction solution stay red.
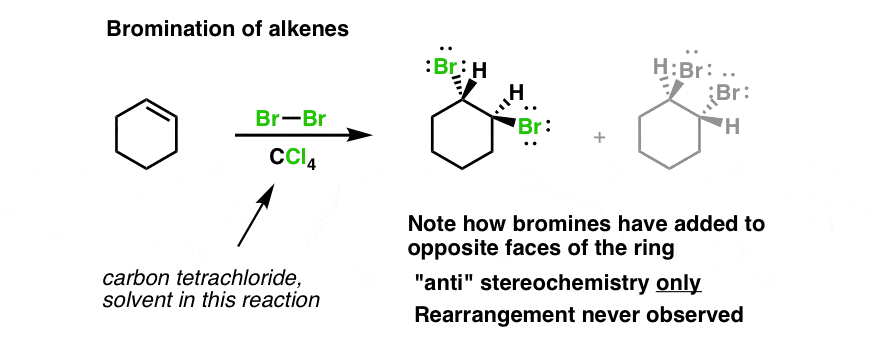
Bromination Of Alkenes Master Organic Chemistry

Bromination Of Cyclohexene Under Conditions Given Below Yields Ltimg Src Https D10lpgp6xz60nq Youtube

Organic Chemistry Product Of Reaction Between Cyclohexene And Bromine In Methanol At 273 K Chemistry Stack Exchange
Reaction Of Bromine With Cyclohexane Cyclohexene And Benzene
0 Comments